Phase Ia (AMD-1)
Safety and Initial Efficacy
- No compound-related safety issues
- Significant improvement in visual acuity over Eylea monotherapy in previously treated patients, demonstrating improved retinal functioning
- Significant anatomical changes, almost completely resolving Intraretinal Fluid (IRF), Subretinal Fluid (SRF), and Pigment Epithelial Detachment (PED), a consequence of angio-regression
- Regression of the pathological vasculature, likely contributing to the long-term effect of the treatment
- Resolution of SHRM, a biomarker for developing fibrosis and atrophy, providing an indication of anti-fibrotic activity
- Significant increase in time between administrations. BCVA at five or six months was as high as any other point in the regimen, suggesting restoration of homeostasis and disease modification
Phase IIa (AMD-2)
Multiple Efficacy Endpoints
Ocugenix has performed trials that have built a compelling story in several critical areas of wet AMD
Fluid Resolution (IRF & SRF)
Vascular regression and reduction of edema/decrease of fluid in active disease
Anti-Fibrotic Activity (SHRM Resolution)
Early elimination of SHRM is supportive
Vascular Regression
BCVA vision improvement well beyond that noted with Eylea in our Phase 1a patients
Extended Treatment-Free Interval (Disease Modification)
CXCR3 signaling restored homeostasis as noted by prolonged disease-free intervals
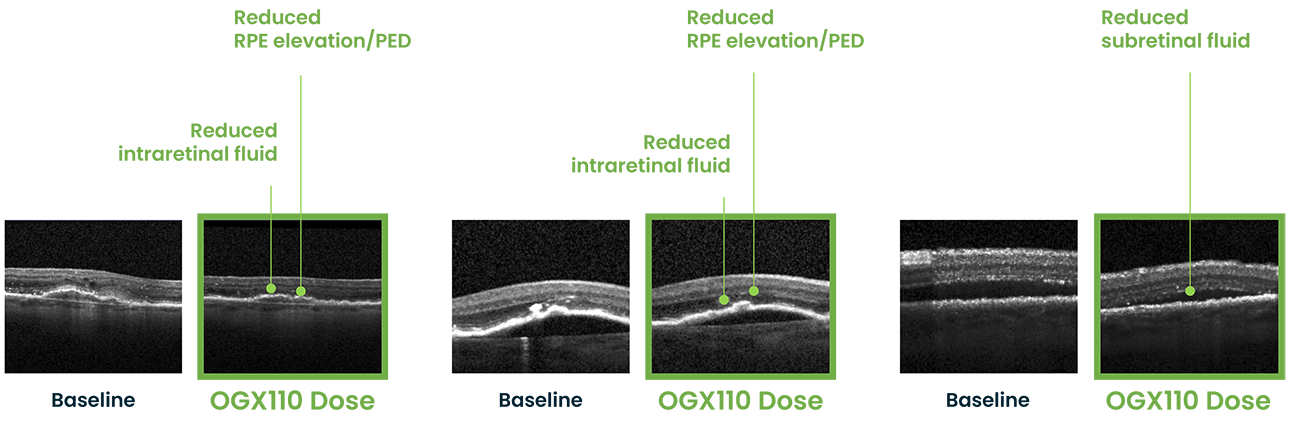
Anatomic evidence that OGX110 decreases fluid after one dose (no anti-VEGFs)
In all three treatment-naïve patients with active exudative AMD, examination of the SD-OCT after a single dose of OGX110 showed resolution of intraretinal fluid, subretinal fluid, and the reduction in pigmented epithelial separation from the underlying eye.
This is consistent with what would be seen as a good response to anti-VEGF therapy. And this is the first demonstration of reduced retinal fluid for an agent other than one that targets VEGF activities.